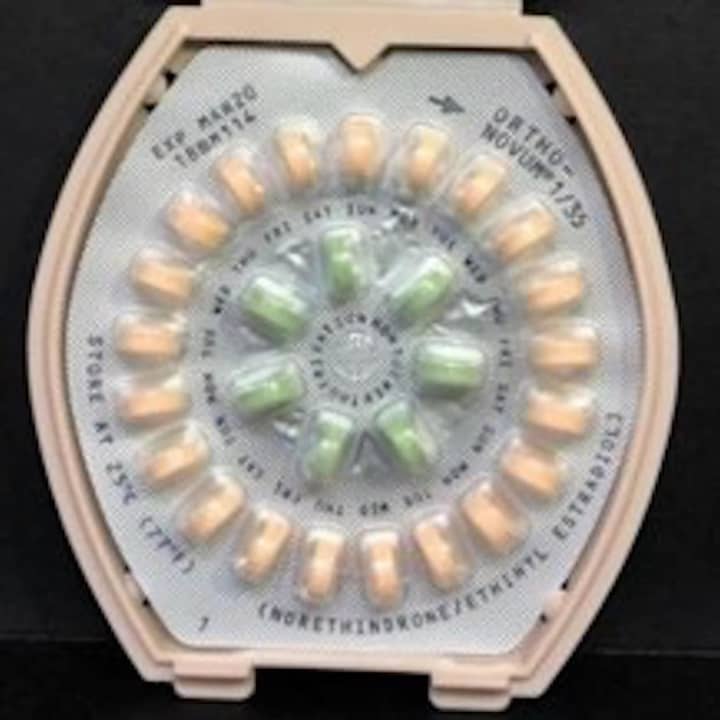
Janssen Pharmaceuticals has initiated a voluntary recall of one lot of Ortho-Novum 1/35 tablets and two lots of Ortho-Novum 7/7/7 tablets to the pharmacy level. According to the FDA, the “patient information provided inside the affected packages does not include the appropriate instructions for the Veridate dispenser.”
“Women should continue to take the 21 ‘active’ pills (with hormones) (peach for Ortho-Novum 1/35; white, light-peach and peach for Ortho-Novum 7/7/7) for three weeks, followed by the one week of green ‘reminder’ pills (without hormones),” the company said in a statement.
The FDA noted that the recall only affects American products, and no other Ortho contraceptive products have been impacted by the recall.
“The potential risk of taking Ortho-Novum without the appropriate instructions for correct use of the Veridate dispenser pack is that the consumer could take the pills in the incorrect order (still receiving an effective dose) or could take an inactive ‘reminder’ pill instead of an ‘active’ pill which could lead to breakthrough bleeding or an unintended pregnancy.”
Click here to follow Daily Voice Fairfield and receive free news updates.